|
It also has a high turnover rate that allows generation of strong signals in a relatively short time span. Horseradish peroxidase is ideal in many respects for these applications because it is smaller, more stable, and less expensive than other popular alternatives such as alkaline phosphatase. Peroxidase, a heme-containing oxidoreductase, is a commercially important enzyme which catalyses the reductive cleavage of hydrogen peroxide by an electron donor. 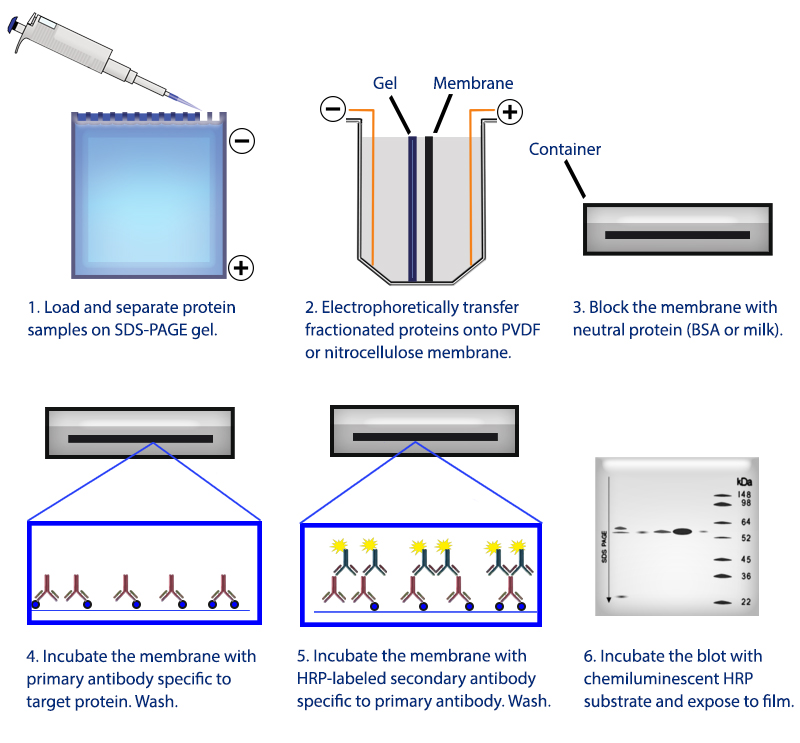
Horseradish peroxidase is also commonly used in techniques such as ELISA and Immunohistochemistry due to its monomeric nature and the ease with which it produces coloured products. Here, the antibody provides the specificity to locate the protein of interest, and the HRP enzyme, in the presence of a substrate, produces a detectable signal. For example, an antibody conjugated to HRP may be used to detect a small amount of a specific protein in a western blot. HRP is often used in conjugates (molecules that have been joined genetically or chemically) to determine the presence of a molecular target. It produces a coloured, fluorimetric, or luminescent derivative of the labeled molecule when incubated with a proper substrate, allowing it to be detected and quantified. Horseradish peroxidase is a 44,173.9-dalton glycoprotein with 6 lysine residues which can be conjugated to a labeled molecule. Luminol is the exception, as it is not a chromophore and light is generated after the HRP-catalyzed reaction. Some of the most common HRP chromogenic substrates. HRP catalyzes the conversion of chromogenic substrates (e.g., TMB, DAB, ABTS) into colored products, and produces light when acting on chemiluminescent substrates (e.g. These substrates fall into several distinct categories. Numerous substrates for horseradish peroxidase have been described and commercialized to exploit the desirable features of HRP. It is a large alpha-helical glycoprotein which binds heme as a redox cofactor.Īlone, the HRP enzyme, or conjugates thereof, is of little value its presence must be made visible using a substrate that, when oxidized by HRP using hydrogen peroxide as the oxidizing agent, yields a characteristic color change that is detectable by spectrophotometric methods. The structure of the enzyme was first solved by X-ray crystallography in 1997 and has since been solved several times with various substrates. 
It catalyzes the oxidation of various organic substrates by hydrogen peroxide. It is a metalloenzyme with many isoforms, of which the most studied type is C. The enzyme horseradish peroxidase ( HRP), found in the roots of horseradish, is used extensively in biochemistry applications.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
